Atomic structure
Structure of atoms
All atoms are composed of a small positively charged nucleus containing protons and neutrons surrounded by a larger negatively charged electron cloud.
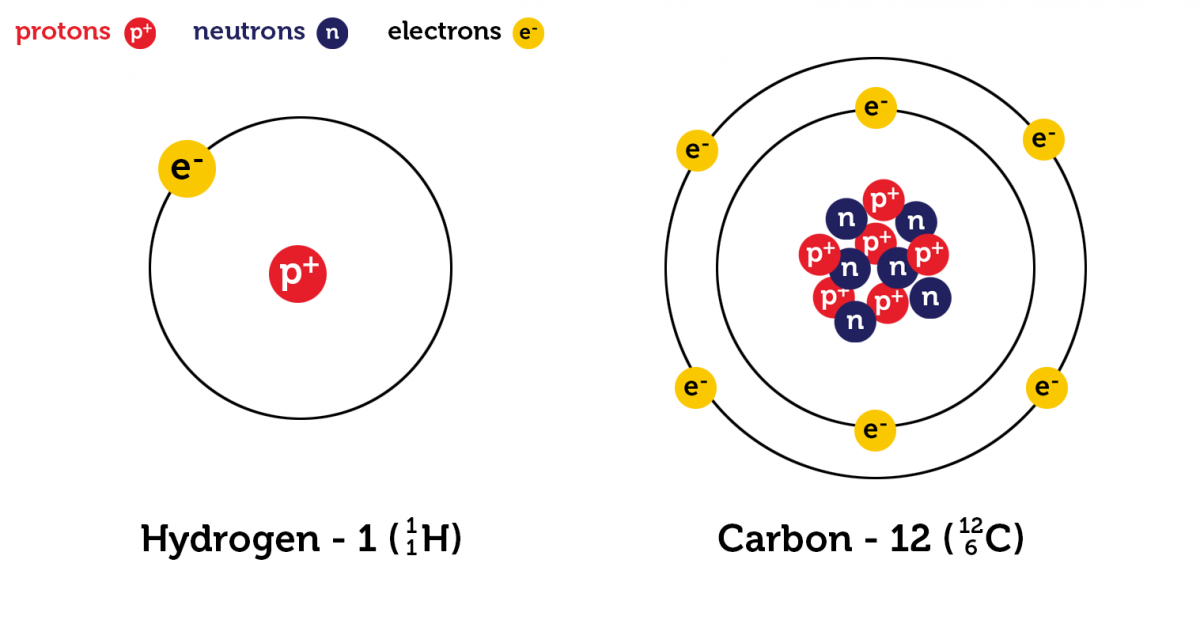
Protons, neutrons and electrons are known as subatomic particles. Properties of the subatomic particles are given in the table below. Subatomic particles present in the nucleus are known as nucleons (e.g. Protons and neutrons). Protons and neutrons are almost identical in mass, whereas electrons are much smaller than the other two. Atoms are uncharged species due to the presence of an equal number of protons and electrons.
Properties of subatomic particles.
| Subatomic particle | Symbol | Charge | Mass (kg) | Size relative to a proton |
|---|---|---|---|---|
| Proton | p | \(+1\) | \(1.673\times10^{-24}\) | \(1\) |
| Neutron | n | \(0\) | \(1.675\times10^{-24}\) | \(1\) |
| Electron | e | \(-1\) | \(9.109\times10^{-28}\) | \(\frac{1}{1800}\) |
Atomic number and mass number
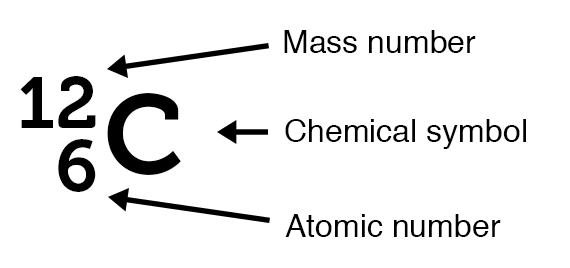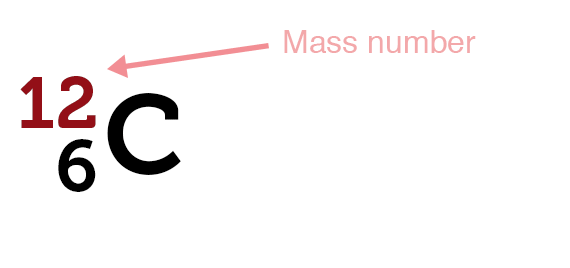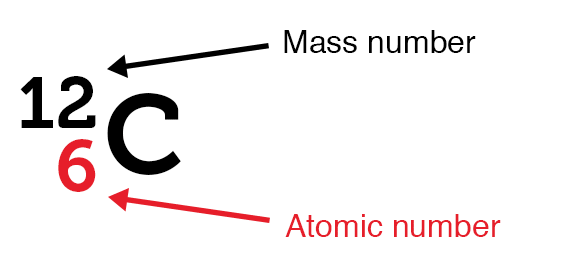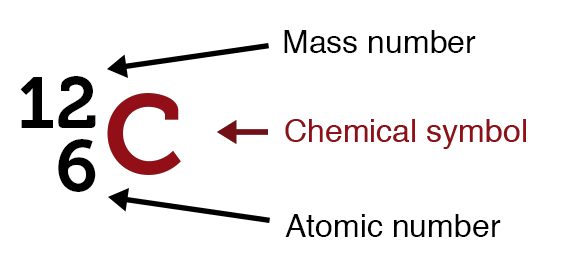 Atomic number and mass number symbols.
Atomic number and mass number symbols. Atomic number
An atomic number (Z) is the number of protons present in the nucleus of an atom. Since an atom contains an equal number of protons and electrons, the atomic number also indicates the number of electrons present in an atom.
- Atomic number (Z) = Number of protons = Number of electrons
Mass number
A mass number (A) is the sum of the number of protons and neutrons present in the nucleus of an atom.
- Mass number (A) = Number of protons + Number of neutrons
The number of subatomic particles present in an atom can be determined using its atomic number and mass number, as shown below.
- Number of protons = Atomic number (Z)
- Number of electrons = Atomic number (Z)
- Number of neutrons = Massnumber (A) - Atomic number (Z)
- Total number of subatomic particles present in an atom = Mass number (A) + Atomic number (Z)
Isotopes and atomic masses
Isotopes are atoms of an element with the same number of protons and electrons but different numbers of neutrons.
Examples:
Carbon has three isotopes: \(\ce{_{6}^{12}C}\), \(\ce{_{6}^{13}C}\), \(\ce{_{6}^{14}C}\)
Chlorine has two isotopes: \(\ce{_{17}^{35}Cl}\), \(\ce{_{17}^{37}Cl}\)
Hydrogen has three isotopes: \(\ce{_{1}^{1}H}\), \(\ce{_{1}^{2}H}\), \(\ce{_{1}^{3}H}\)
Atomic mass is the weighted average mass of an atom of an element. Relative percentage abundancy and atomic mass of each isotope are considered when determining an element’s atomic mass.

Build an atom with the simulation below or open this activity in a new window.
