The Electron - an overview
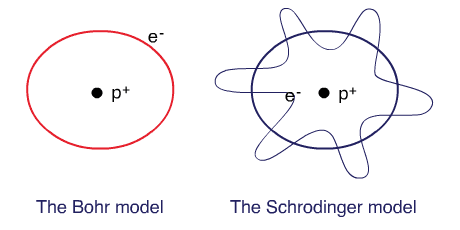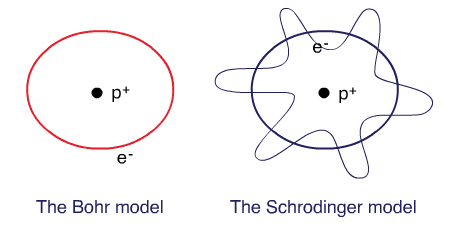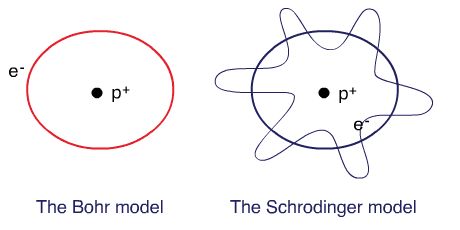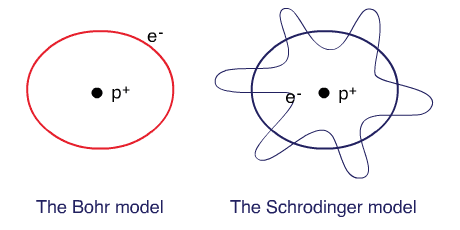
Bohr planetary (particle) model
Electrons are tiny particles that are revolving around the nucleus in circular orbits similar to the solar system.
Schrodinger’s wave mechanical model
Electrons behave as waves around the nucleus such that a stable energy state is only established when an integral number of wavelengths are “quantized” (quantum means a specific amount) into the orbital to produce a “stationary state”, a confined electron wave. In this model, electrons occupy a three-dimensional space around the nucleus.
The Bohr model and the Schrodinger’s model:
Energy levels within an atom
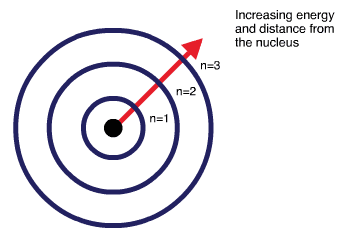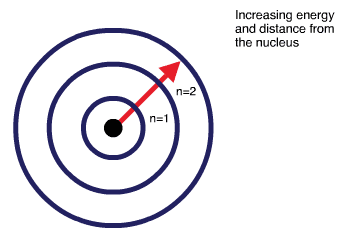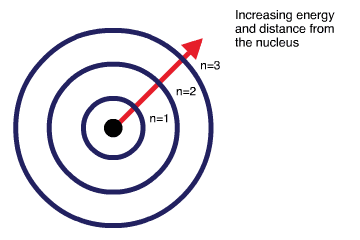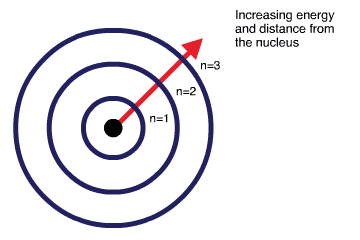
Space, where electrons move around the nucleus, is divided into subspaces known as shells, subshells and orbitals.
- Electron shells - Electron shells are named outward from the nucleus as \(1,2,3,4\) and so on. The energy of the electron shells increases with the distance from the nucleus. Therefore, the electrons closest to the nucleus have lower energy than the electrons located further away.
- Electron subshells - The number of subshells in a shell is equal to the shell number. Subshells are labeled as \(s,p,d\) and \(f\). Each subshell contains a certain number of electrons. An \(s\) subshell contains up to \(2\) electrons, a \(p\) subshell contains up to \(6\) electrons, a \(d\) subshell contains up to \(10\) electrons, and a \(f\) subshell contains up to \(14\) electrons.
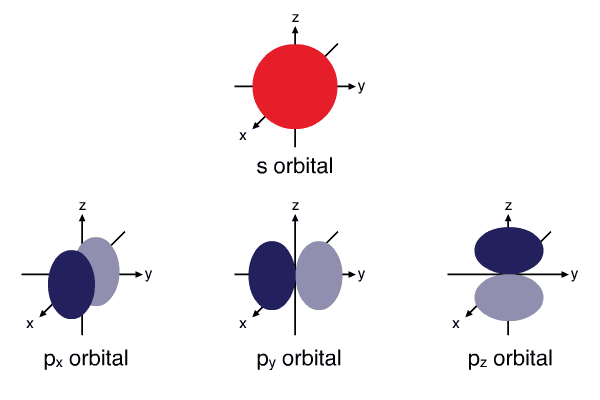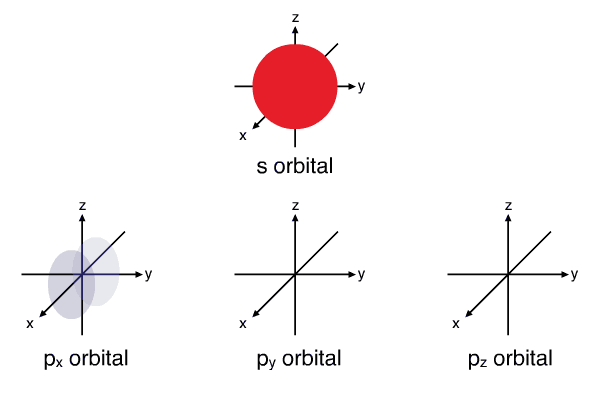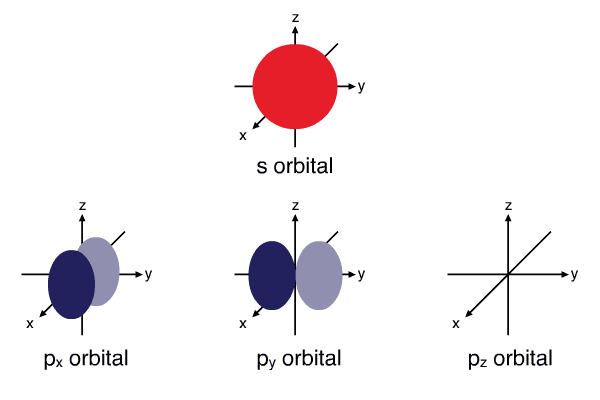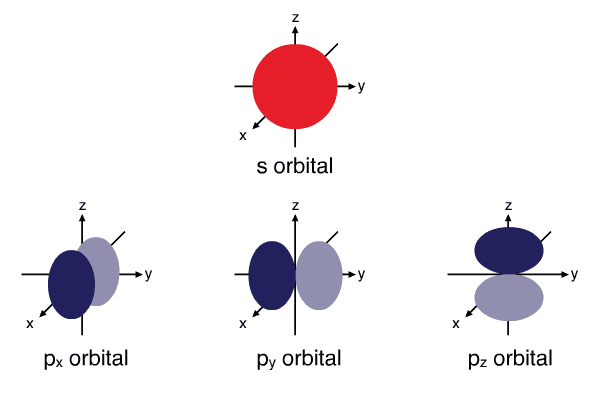
- Electron orbitals - An electron orbital can contain a maximum of two electrons where one must spin up and the other spin down. Therefore, an \(s\) subshell has one orbital, a \(p\) subshell has three orbitals, a \(d\) subshell has five orbitals and an \(f\) subshell has seven orbitals. Orbitals have distinct shapes, \(s\) orbitals are spherical, \(p\) orbitals are dumb-bell. Orbitals within the same subshell that have the same shape are generally of equal energy but have a different orientation in space, such as \(p_{x},p_{y},p_{z}\) (\(x,y,z\) denote the orientation in space).
Writing electron configurations
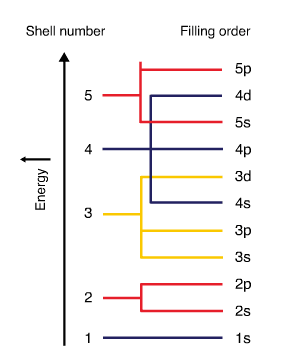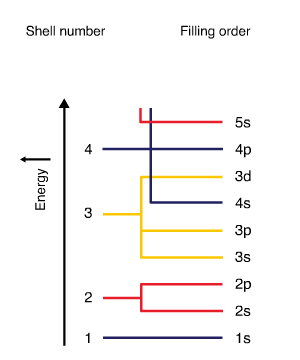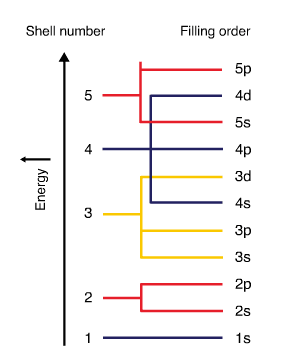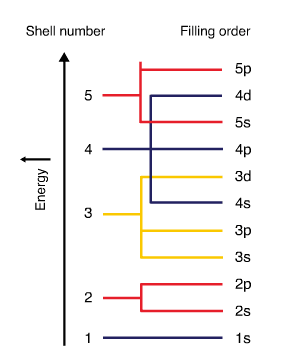
- Electron subshells are filled in order of increasing energy. The figure below displays the electron filling order. Subshell \(4s\) has lower energy than subshell \(3d\). Therefore \(4s\) fills before \(3d\).
- Each orbital can accommodate up to \(2\) electrons.
- Electrons fill into orbitals of a subshell so that each orbital acquires one electron before any orbital acquires a second electron. All single electrons must have the same spin.
Shapes of atomic orbitals:
The electron configuration of selected elements
| Element | Atomic number | Electron configuration |
|---|---|---|
| \(\ce{H}\) | \(1\) | \(1s^{1}\) |
| \(\ce{He}\) | \(2\) | \(1s^{2}\) |
| \(\ce{Li}\) | \(3\) | \(1s^{2}\,2s^{1}\) |
| \(\ce{Be}\) | \(4\) | \(1s^{2}\,2s^{2}\) |
| \(\ce{B}\) | \(5\) | \(1s^{2}\,2s^{2}2p^{1}\) |
| \(\ce{C}\) | \(6\) | \(1s^{2}\,2s^{2}2p^{2}\) |
| \(\ce{N}\) | \(7\) | \(1s^{2}\,2s^{2}2p^{3}\) |
| \(\ce{O}\) | \(8\) | \(1s^{2}\,2s^{2}2p^{4}\) |
| \(\ce{F}\) | \(9\) | \(1s^{2}\,2s^{2}2p^{5}\) |
| \(\ce{Ne}\) | \(10\) | \(1s^{2}\,2s^{2}2p^{6}\) |
