Atoms, molecules and compounds
\[\require{mhchem}\]
Atoms are the basic building blocks of elements that still have the properties of the element. They are too small to be observed by the naked eye.
Examples: \(\ce{H\;He\;C\;Mg\;Fe}\)
Elements are the building blocks of all matter and composed of one type of atom. Some of the elements exist as molecules, for instance, \(\ce{S8\;O2\;Cl2}\)
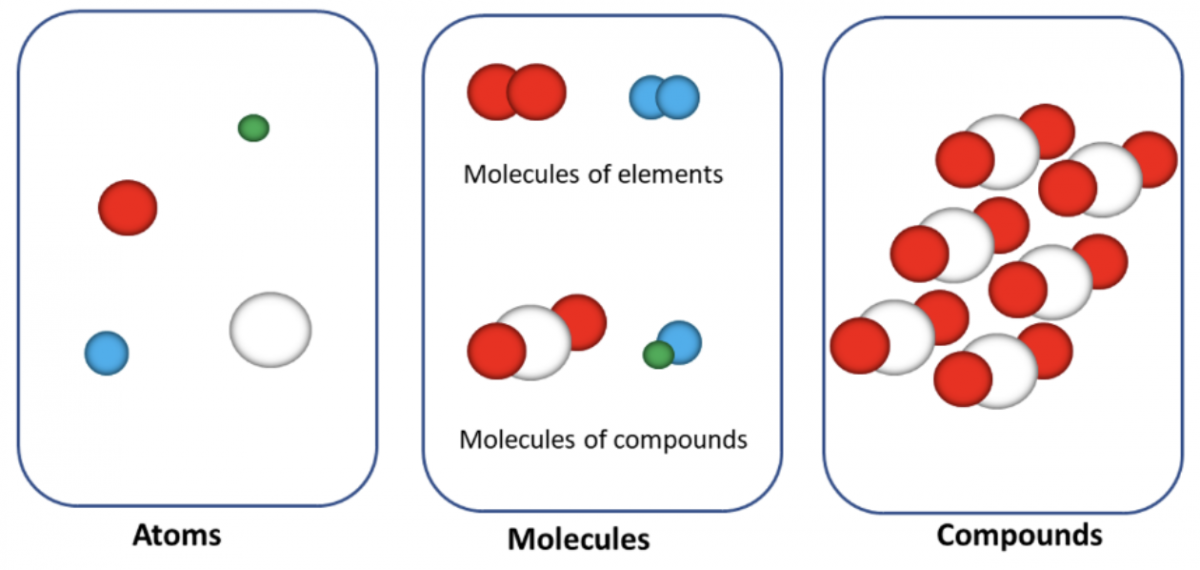
Molecules and Compounds
A molecule is a group of two or more atoms combined chemically and functions as a unit. The simplest molecule that can exist is a diatomic molecule that contains two atoms such as \(\ce{O2}\) and \(\ce{H2}\).
- Compounds contain different types of atoms in fixed proportions.
- Compounds and molecules are uncharged species.
Examples of molecules and compounds
| Molecules | Compounds |
|---|---|
| \(\ce{H2}\) | \(\ce{H2O}\) |
| \(\ce{O3}\) | \(\ce{CO2}\) |
| \(\ce{S8}\) | \(\ce{C2H5OH}\) |
| \(\ce{CO2}\) | \(\ce{CH3COOH}\) |
| \(\ce{H2O}\) | \(\ce{FeSO4}\) |
Element symbols
Each element has a unique name and a chemical symbol. Examples of element symbols are given in the table below. All the discovered elements are tabulated in the modern periodic table.
Names and symbols of the elements
| Element | Chemical symbol |
|---|---|
| Hydrogen | \(\ce{H}\) |
| Carbon | \(\ce{C}\) |
| Calcium | \(\ce{Ca}\) |
| Silver | \(\ce{Ag}\) |
| Copper | \(\ce{Cu}\) |
Chemical formulas
Chemical formulas represent the chemical composition of molecules and compounds. They consist of chemical symbols of the elements present in the compound and numerical subscript, which demonstrates the number of atoms of each element involved in the formation of the compound.
Examples:
- The chemical formula for the compound sucrose is \(\ce{C12H22O11}\)
This formula shows that sucrose contains three different elements: \(\ce{C\;H\;O}\)
The total number of atoms: 45
The number of each type of atom: \(\ce{C}\) \(-12\), \(\ce{H}\) \(-22\), \(\ce{O}\) \(-11\) - The chemical formula for the compound Calcium phosphate is \(\ce{Ca3(PO4)2}\)
This formula shows that one unit of Calcium phosphate is composed of three different elements : \(\ce{Ca\;P\;O}\)
The total number of atoms: \(13\)
The number of each type of atom: \(\ce{Ca}\) \(-3\), \(\ce{P}\) \(-2\), \(\ce{O}\) \(-8\)(For \(\ce{O}\) and \(\ce{P}\) multiply the subscript number by the number written after the parenthesis.)
Test your knowledge - take the quiz>
Use the interactives below to make your own molecule or create an isotope - or got the site by selecting "PHET website" from the bottom right hand corner
