.
Lewis structure exercises
\[\require{mhchem}\]
Practise drawing the Lewis structure of molecules using the exercises below. Select answers to see the correct drawings.
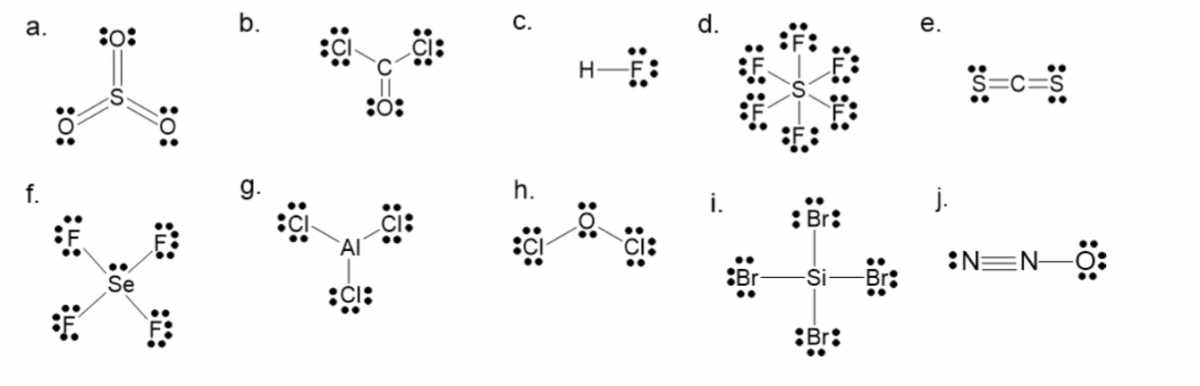
- Draw the Lewis structures of the following molecules.
- \(\ce{SO}_{3}\)
- \(\ce{COCl}_{2}\)
- \(\ce{HF}\)
- \(\ce{SF}_{6}\)
- \(\ce{CS}_{2}\)
- \(\ce{SeF}_{4}\)
- \(\ce{AlCl}_{3}\)
- \(\ce{Cl}_{2}\ce{O}\)
- \(\ce{SiBr}_{4}\)
- \(\ce{N}_{2}\ce{O}\)
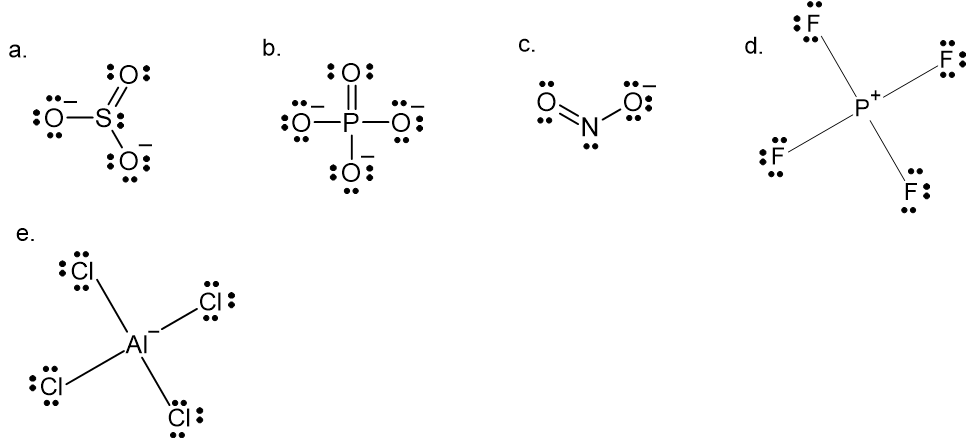
2. Draw the Lewis structures of the following polyatomic ions
- \(\ce{SO}_{3}^{2-}\)
- \(\ce{PO}_{4}^{3-}\)
- \(\ce{NO}_{2}^{-}\)
- \(\ce{PF}_{4}^{+}\)
- \(\ce{AlCl}_{4}^{-}\)