Chemical bonding quiz
\[\require{mhchem}\]
Test your knowledge of ionic bonding here. 5 example questions with explanations and 25 quiz questions to challenge your knowledge.
Worked Exercises
1. Determine how many electrons an aluminium atom has to lose or gain to obtain a noble gas electron configuration.
Answer
The purpose of losing or gaining an electron is to acquire an octet in the valence shell. \(\ce{Al}\)’s electron configuration is \(1s^{2}2s^{2}2p^{6}3s^{2}3p^{1}\). The valence shell contains three electrons. \(\ce{Al}\) can obtain the electron configuration of \(\ce{Ne}\), which is the noble gas just before \(\ce{Al}\), by losing those three electrons, . The \(\ce{Al}\) atom has \(13\) electrons and \(13\) protons in the neutral state. When it loses three electrons, \(\ce{Al}\) has three excess protons giving rise to a cation, \(\ce{Al}^{3+}\). \[ \ce{Al}^{3+}\;1s^{2}2s^{2}2p^{6}=\left[\ce{Ne}\right] \]
2. The neutral \(\ce{O}\) atom has eight electrons and eights protons. Explain how \(\ce{O}\) atom acquires valence shell octet.
Answer
The electron configuration of \(\ce{O}\) is \(1s^{2}2s^{2}2p^{4}\). The valence shell has four electrons. It can achieve an octet and stable noble gas electron configuration by gaining two electrons into a \(2p\) orbital. Therefore, \(\ce{O}\) produces \(\ce{O}^{2-}\) ion, which has two excess electrons. \[ \ce{O}^{2-}\;1s^{2}2s^{2}2p^{6}=\left[\ce{Ne}\right] \]
3. Determine the chemical formula of the compound produced when \(\ce{Ca}^{2+}\) and \(\ce{F}^{-}\) ions interact.
Answer
There are two ways of writing chemical formulas.
Method 1:
First, write down the symbol of the cation with its charge followed by the anion.
\[ \ce{Ca}^{2+} \] \[ \ce{Ca}^{2+}\ce{F}^{-} \] Next, cross each ions charge (only the numerical value, not the sign) and write it down as subscripts. So, calcium ion’s charge will be written next to fluoride ion, and fluoride ion’s charge will be written next to calcium ion, in both cases as subscripts
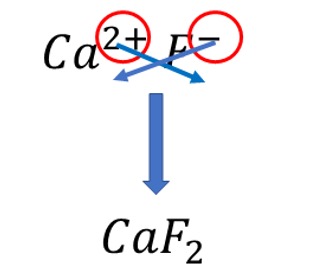
Method 2:
\(\ce{Ca}\) ion has a \(+2\) charge, and \(\ce{F}\) ion has a \(-1\) charge. Two \(\ce{F}\) ions, each with \(-1\) charge, will neutralise the \(+2\) charge of \(\ce{Ca}\) ion. Therefore, the chemical formula is \[ \ce{CaF2} \]
4. Write the chemical formula for a compound composed of \(\ce{Al}^{3+}\) and \(\ce{O}^{2-}\).
Answer
First, write down the symbol of the cation followed by the anion with their charges. \[ \ce{Al}^{3+}\ce{O}^{2-} \] Next, cross over the numerical values of the charges to get the chemical formula. \[ \ce{Al}_{2}\ce{O}_{3} \]
5. Write the chemical formula of the compound formed by copper\(\left(\mathrm{II}\right)\) ions and sulfate ions.
Answer
Copper\(\left(\mathrm{II}\right)\) ion \(=\ce{Cu}^{2+}\)
Sulfate ion \(=\ce{SO}_{4}^{2-}\)
Copper ion has a \(+2\) charge, and sulphate ion has a \(-2\) charge. The \(+2\) charge of copper ion cancels off to the \(-2\) charge of sulphate ion resulting in a neutral compound.
So, the chemical formula is \(\ce{CuSO}_{4}\).
Exercises
- \(\ce{Ca}\) loses two electrons to acquire the electron configuration of \(\ce{Ar}\). What is the chemical symbol of the ion that it forms?
- \(\ce{Ca}^{2-}\)
- \(\ce{Ca}^{2+}\)
- \(\ce{Ca}\)
- \(\ce{Ca}^{+}\)
b \(\ce{Ca}^{2+}\)
- When \(\ce{N}\) reacts with other elements, it gains three electrons. What is the chemical symbol of the resulting ion?
- \(\ce{N}^{3-}\)
- \(\ce{N}^{3+}\)
- \(\ce{N}\)
- \(\ce{N}_{2}\)
- \(\ce{N}^{3-}\)
- Which of the following statements is true regarding ions?
- Cations are formed when metals gain electrons.
- Anions are formed when neutral atoms gain electrons.
- By losing electrons, alkaline earth metals acquire the electron configuration of a noble gas just after them.
- By gaining electrons, halogens achieve the electron configuration of a noble gas just before them.
b. Anions are formed when neutral atoms gain electrons.
- Which of the following statements is incorrect?
- Metals tend to form cations
- Nonmetals tend to form anions
- Octet rule states that atoms lose or gain electrons to acquire completely filled valence shell that contains \(8\) electrons
- Noble gases are very reactive due to their electron configurations
d. Noble gases are very reactive due to their electron configurations
- What is the electron configuration of \(\ce{Mg}^{2+}\)?
- \(1s^{2}2s^{2}2p^{6}\)
- \(1s^{2}2s^{2}2p^{6}3s^{2}\)
- \(1s^{2}2s^{2}2p^{6}3s^{2}3p^{6}\)
- \(1s^{2}2s^{2}2p^{6}3s^{2}3p^{6}4s^{2}\)
a. \(1s^{2}2s^{2}2p^{6}\)
- What is the electron configuration of \(\ce{F}^{-}\)?
- \(1s^{2}2s^{2}2p^{5}\)
- \(1s^{2}2s^{2}2p^{6}\)
- \(1s^{2}2s^{2}2p^{6}3s^{2}3p^{6}\)
- \(1s^{2}\)
b. \(1s^{2}2s^{2}2p^{6}\)
- What is the electron configuration of the ion that \(_{19}\ce{K}\) forms?
- \(1s^{2}2s^{2}2p^{6}3s^{2}3p^{6}4s^{1}\)
- \(1s^{2}2s^{2}2p^{6}3s^{2}3p^{6}\)
- \(1s^{2}2s^{2}2p^{6}\)
- \(1s^{2}2s^{2}2p^{6}3s^{2}3p^{6}4s^{2}4p^{6}\)
b. \(1s^{2}2s^{2}2p^{6}3s^{2}3p^{6}\)
- How many electrons does \(\ce{O}\) gain to acquire a noble gas configuration?
- \(8\)
- \(6\)
- \(2\)
- \(10\)
c. \(2\)
- Which of the following ions does exist?
- \(\ce{S}^{-}\)
- \(\ce{C}^{2+}\)
- \(\ce{Al}^{4+}\)
- \(\ce{Cs}^{+}\)
d. \(\ce{Cs}^{+}\)
- What are the common ions formed by \(_{29}\ce{Cu}\)?
- \(\ce{Cu}^{+}\)and \(\ce{Cu}^{2+}\)
- \(\ce{Cu}^{+}\)and \(\ce{Cu}^{3+}\)
- \(\ce{Cu}^{2+}\)and \(\ce{Cu}^{3+}\)
- \(\ce{Cu}^{+}\), \(\ce{Cu}^{2+}\)and \(\ce{Cu}^{3+}\)
a. \(\ce{Cu}^{+}\)and \(\ce{Cu}^{2+}\)
- What is the symbol of the ion formed by Beryllium losing two electrons from the valence shell?
- \(\ce{Be}^{+}\)
- \(\ce{Be}^{2+}\)
- \(\ce{Ba}^{2+}\)
- \(\ce{Ba}^{+}\)
b. \(\ce{Be}^{2+}\)
- What is the name of \(\ce{Cr}^{2+}\) ion?
- Chromium\(\left(\mathrm{II}\right)\)
- Chromous
- Chromic
- a and b
d. a and b
- Which of the following combinations correctly name the given ions?
\(\ce{Br}^{-}\), \(\ce{Al}^{3+}\), \(\ce{Fe}^{3+}\)
- Bromide, Aluminum\(\left(\mathrm{III}\right)\), Ferric
- Bromium ion, Aluminum ion, Iron
- Bromide ion, Aluminum ion, Iron\(\left(\mathrm{III}\right)\)
- Bromide, Aluminum, Ferrous
c. Bromide ion, Aluminum ion, Iron\(\left(\mathrm{III}\right)\)
- What is the name of \(\ce{H}_{3}\ce{O}^{+}\)ion?
- Hydroxide ion
- Hydronium ion
- Hydrogen peroxide ion
- Hydrogen ion
b. Hydronium ion
- Which of the following options incorrectly named the given polyatomic ion?
- \(\ce{CO}_{3}^{2-}\) \(=\) Carbonate ion
- \(\ce{CH}_{3}\ce{COO}^{-}\) \(=\) Acetate ion
- \(\ce{CN}^{-}\) \(=\)Cyanide ion
- \(\ce{MnO}_{4}^{-}\)= Manganese ion
d. \(\ce{MnO}_{4}^{-}\)= Manganese ion
- What is the formula of the compound formed by \(\ce{NH}_{4}^{+}\) and \(\ce{Cl}^{-}\) ions?
- \(\ce{NH}_{4}\ce{Cl}\)
- \(\ce{NHCl}_{4}\)
- \(\ce{ClNH}_{4}\)
- \(\ce{NHCl}\)
a. \(\ce{NH}_{4}\ce{Cl}\)
- What is the chemical formula for sodium hydrogen carbonate?
- \(\ce{NaCO}_{3}\)
- \(\ce{NaHCO}_{3}\)
- \(\ce{Na}_{2}\ce{CO}_{3}\)
- \(\ce{NaHC}\)
b. \(\ce{NaHCO}_{3}\)
- What is the chemical formula for calcium carbonate?
- \(\ce{CaCO}_{3}\)
- \(\ce{CaCO}\)
- \(\ce{CaO}\)
- \(\ce{CaHCO}_{3}\)
a. \(\ce{CaCO}_{3}\)
- Which of the following options matches the name and the chemical formula
- Ammonium carbonate - \(\left(\ce{NH}_{4}\right)_{2}\ce{CO}_{3}\)
- Magnesium hydroxide - \(\ce{MgOH}\)
- Silver nitrate - \(\ce{AgNO}\)
- Sodium hypochlorite - \(\ce{NaClO}\)
a. Ammonium carbonate - \(\left(\ce{NH}_{4}\right)_{2}\ce{CO}_{3}\)
- Which of the following options gives the correct name for \(\ce{AuCl}_{3}\)?
- Gold chloride
- Gold chloride\(\left(\mathrm{III}\right)\)
- Gold\(\left(\mathrm{III}\right)\) chloride
- Gold\(\left(\mathrm{III}\right)\) chloride\(\left(\mathrm{III}\right)\)
c. Gold\(\left(\mathrm{III}\right)\) chloride
- What is the charge of silver ion in \(\ce{Ag}_{2}\ce{S}\)?
- \(+2\)
- \(-2\)
- \(+1\)
- \(-1\)
c. \(+1\)
- Which of the following statements is true regarding the charge of the cation of the given compounds?
| Compound | Charge of the cation |
|---|---|
| \(\ce{Fe}_{2}\ce{O}_{3}\) | \(x\) |
| \(\ce{Fe}\left(\ce{NO}_{3}\right)_{2}\) | \(y\) |
| \(\ce{Fe}_{2}\left(\ce{SO}_{4}\right)_{3}\) | \(z\) |
- \(x=+3,y=+2,z=+3\)
- \(x=+2,y=+2,z=+3\)
- \(x=+1,y=+2,z=+3\)
- \(x=+2,y=+2,z=+2\)
a. \(x=+3,y=+2,z=+3\)
- Which of the following statements is correct?
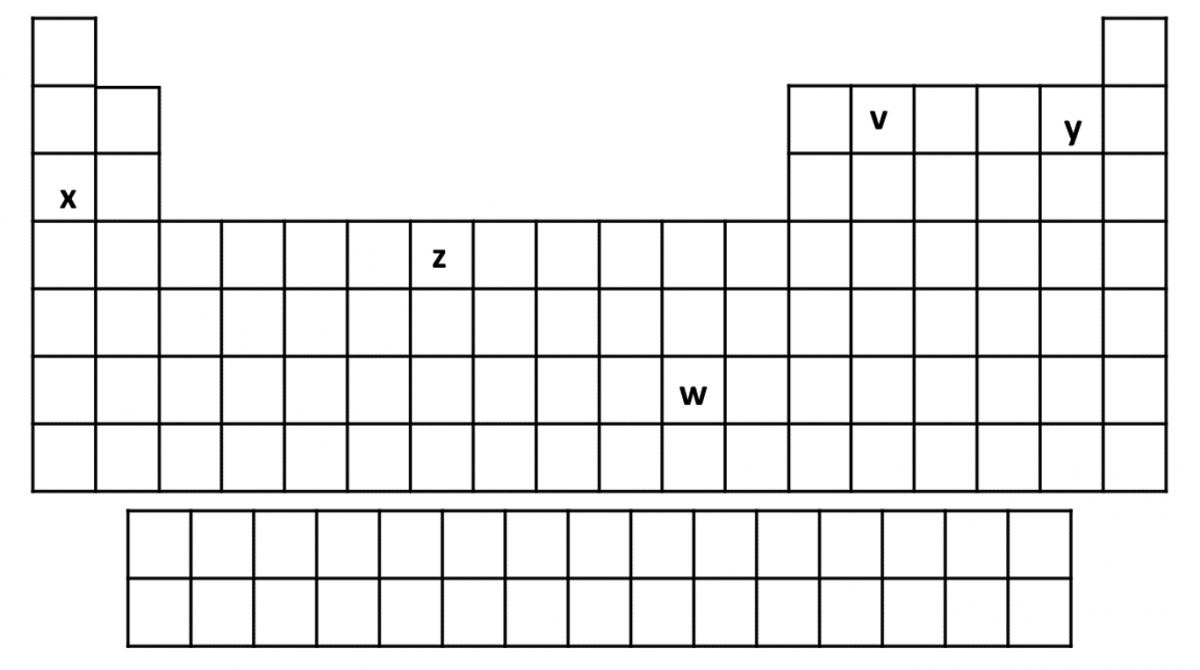
- Element \(x\) only forms \(+1\) ions
- Element y only forms \(-1\) ions
- Element \(v\) does not form ions
- Element \(z\) and \(w\) form multiple ions
- Only 1 and 2
- Only 3 and 4
- all above
- only 1
c. all above
- Components of four different ionic compounds are shown in the following periodic table. Components of the same compound are shown in the same colour. What are the likely chemical formulas for each of these compounds?
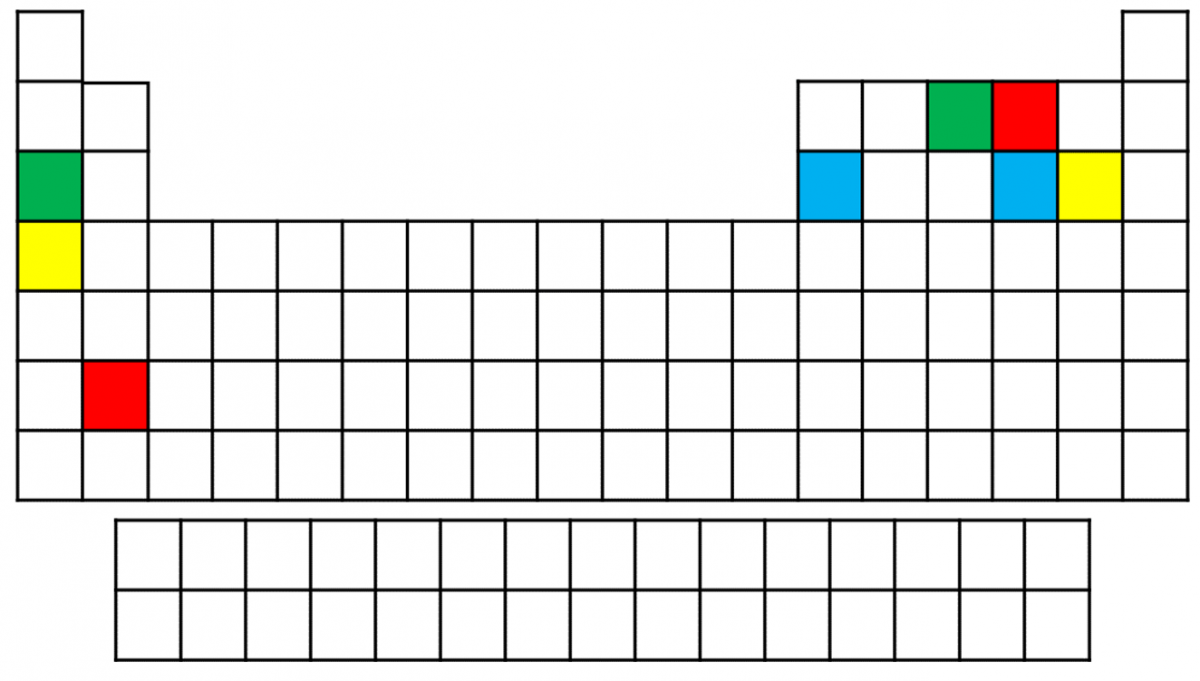
- Green -\(\ce{Na}_{3}\ce{N}\), Yellow - \(\ce{KCl}\), Red - \(\ce{BaO}\), Blue - \(\ce{Al}_{2}\ce{S}_{3}\)
- Green -\(\ce{NaNO}_{3}\), Yellow - \(\ce{KCl}\), Red - \(\ce{BaO}\), Blue - \(\ce{Al}_{2}\ce{SO}_{4}\)
- Green -\(\ce{Na}\ce{N}\), Yellow - \(\ce{KCl}\), Red - \(\ce{BaO}_{2}\), Blue - \(\ce{Al}_{2}\ce{S}_{3}\)
- Green -\(\ce{Na}_{3}\ce{N}\), Yellow - \(\ce{KCl}_{2}\), Red - \(\ce{BaO}\), Blue - \(\ce{Al}_{2}\ce{S}_{3}\)
a. Green -\(\ce{Na}_{3}\ce{N}\), Yellow - \(\ce{KCl}\), Red - \(\ce{BaO}\), Blue - \(\ce{Al}_{2}\ce{S}_{3}\)
- Which of the following options has isoelectronic species?
- \(\ce{Mg}^{2+}\), \(\ce{Ne}\), \(\ce{Na}^{+}\), \(\ce{F}^{-}\)
- \(\ce{Ne}\), \(\ce{K}^{+}\), \(\ce{Na}^{+}\), \(\ce{O}^{2-}\)
- \(\ce{Al}^{3+}\), \(\ce{He}\), \(\ce{Ne}\), \(\ce{Ar}\)
- \(\ce{Mg}^{2+}\), \(\ce{Ar}\), \(\ce{Ca}^{2+}\), \(\ce{K}^{+}\)
a. \(\ce{Mg}^{2+}\), \(\ce{Ne}\), \(\ce{Na}^{+}\), \(\ce{F}^{-}\)
