Periodic table quiz
Take this quiz to test your knowledge of the periodic table
Worked exercises
How many valence electrons does each of the following atoms have: (a) \(\ce{P}\) (b) \(\ce{Sc}\) (c) \(\ce{F}\) (d) \(\ce{Ca}\) (e) \(\ce{Fe}\)?
Answer
The group numbers of these elements can be used to determine the number of valence electrons. For groups \(1-10\), group number equals to the number of valence electrons. For groups \(12-18\), subtract \(10\) out of the group number to get the valence electrons. For group \(11\), the number of valence electrons can be anticipated as \(1\) due to the \(s^{1}\) electron configuration. However, there are a few exceptions in this case.
| Element | Group number | Valence electrons |
|---|---|---|
| \(\ce{P}\) | \(15\) | \(15-10=5\) |
| \(\ce{Sc}\) | \(3\) | \(3\) |
| \(\ce{F}\) | \(17\) | \(17-10=7\) |
| \(\ce{Ca}\) | \(2\) | \(2\) |
| \(\ce{Fe}\) | \(8\) | \(8\) |
Predict which atom is larger for the following pairs of atoms:
(a) \(\ce{C}\) and \(\ce{F}\)
(b) \(\ce{Na}\) and \(\ce{Cs}\)
(c) \(\ce{Sc}\) and \(\ce{Ni}\)
Answer
Each atom’s position in the periodic table and atomic number can be used to predict its atomic size.
(a) \(\ce{C}\) (\(Z=6\)) and \(\ce{F}\) (\(Z=9\)) are located in the second period. When moving across a period from left to right atomic radii decrease as a result of increasing Z. \(\ce{C}\) lies to the left of \(\ce{F}\) in the same row. Therefore, \(\ce{C}\) is larger than \(\ce{F}\).
(b) \(\ce{Na}\) (\(Z=11\)) and \(\ce{Cs}\) (\(Z=55\)) are located in group 1. Atomic radii increase from top to bottom of the periodic table as \(n\) increases. \(\ce{Cs}\) is located below \(\ce{Na}\) in the same column. Thus, \(\ce{Cs}\) has a larger radius than \(\ce{Na}\).
(c) \(\ce{Sc}\) (\(Z=21\)) and \(\ce{Ni}\) (\(Z=28\)) are located in the same row of the \(d\) block. Across \(d\) block and \(f\) block, atomic radii change is not significant. Therefore, \(\ce{Sc}\) and \(\ce{Ni}\) approximately have the same radius.
Exercises
- Which of the following set of elements possess similar chemical properties?
- \(\ce{_{19}K}\) and \(\ce{_{29}Cu}\)
- \(\ce{_{13}Al}\) and \(\ce{_{14}Si}\)
- \(_{17}\ce{Cl}\) and \(\ce{_{53}I}\)
- \(\ce{_{11}Na}\) and \(\ce{_{12}Mg}\)
- Which of the following statements is true regarding the periodic table?
- The horizontal rows in the periodic table are known as groups
- The verticle columns in the periodic table are known as periods
- In the periodic table, when the atomic number increases, the properties of the elements repeat regularly
- The periodic table is classified into blocks based on the electron filling pattern into shells
- Which of the following sets of elements are metals?
- \(\ce{_{1}H}\) and \(\ce{_{3}Li}\)
- \(\ce{_{26}Fe}\) and \(\ce{_{29}Cu}\)
- \(\ce{_{8}O}\) and \(\ce{_{16}S}\)
- \(\ce{_{5}B}\) and \(\ce{_{14}Si}\)
- Which of the following sets of elements are nonmetals?
- \(\ce{_{2}He}\) and \(\ce{_{17}Cl}\)
- \(\ce{_{1}H}\) and \(\ce{_{20}Ca}\)
- \(\ce{_{14}Si}\) and \(\ce{_{33}As}\)
- \(\ce{_{11}Na}\) and \(\ce{_{12}Mg}\)
- Which of the following elements is a metalloid?
- \(\ce{_{7}N}\)
- \(\ce{_{14}Si}\)
- \(\ce{_{27}Co}\)
- \(\ce{_{53}I}\)
- Which of the following properties are unique to metals?
- Little or no metallic lustre (dull appearance)
- Poor conductors of electricity and heat
- Solids under room temperature
- Low melting points
- The element highlighted in red in the given periodic table is a
- Alkali metal
- Alkaline earth metal
- Halogen
- Noble gas
- Element 4 in the given periodic table is a?
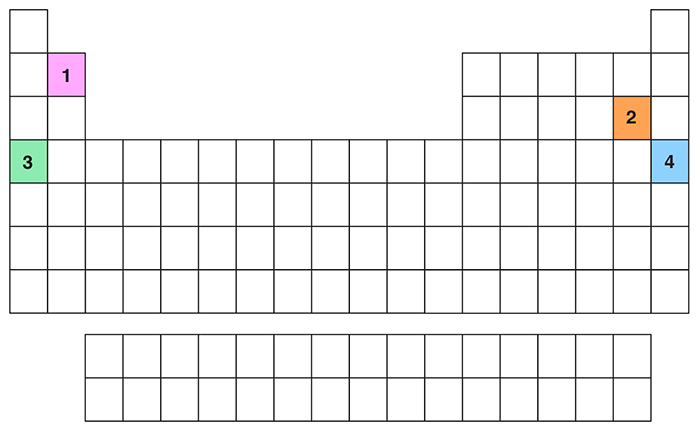
- Alkali metal
- Alkaline earth metal
- Halogen
- Noble gas
- Which of the highlighted elements in the given periodic table is an alkali metal as well as an \(s\) block element?

- Element 1
- Element 3
- Element 4
- Element 2
- Which of the following statements is true regarding the highlighted elements in the given periodic table?

- Element 2 is a halogen as well as a \(p\) block element
- Element 1 is a noble gas
- Element 4 is a halogen as well as a \(p\) block element
- Elements 1 and 3 belongs to the \(d\) block
- Which of the following statements classify the given electron configurations correctly?
- \(1s^{2}\,2s^{2}2p^{6}\) - \(d\) block element
- \(1s^{2}\,2s^{2}2p^{6}\,3s^{2}\)- \(p\) block element
- \(1s^{2}\,2s^{2}2p^{6}\,3s^{2}3p^{6}3d^{2}\,4s^{2}\)- \(f\) block element
- \(1s^{2}\,2s^{1}\)- \(s\) block element
- Which of the following statements is incorrect?
- Representative elements are found in \(s\) area and the first five columns of \(p\) area
- Transition elements are found in \(d\) area, and they are all metals
- Inner transition elements are found in \(f\) area, and they are all nonmetals
- Noble gases are found in the last column of the \(p\) area, and they are all nonmetals
- How many valence electrons does \(\ce{_{11}Na}\) have?
- \(1\)
- \(11\)
- \(9\)
- \(3\)
- How many valence electrons does Group \(13\) have?
- \(13\)
- \(3\)
- \(5\)
- \(10\)
- How many valence electrons does \(\ce{_{27}Co}\) have?
- \(1\)
- \(6\)
- \(9\)
- \(10\)
- Which of the following atoms has the largest atomic radius?
- \(\ce{C}\)
- \(\ce{F}\)
- \(\ce{Be}\)
- \(\ce{Mg}\)
- Which of the following pairS of atoms has nearly the same size?
- \(\ce{Li}\) and \(\ce{K}\)
- \(\ce{Al}\) and \(\ce{Cl}\)
- \(\ce{Ne}\) and \(\ce{Kr}\)
- \(\ce{Cr}\) and \(\ce{Co}\)
- Arrange the following atoms in order of increasing first ionisation energy.
\(\ce{S}\), \(\ce{Ar}\), \(\ce{$\ce{Mg}$ }\), \(\ce{Sr}\) and \(\ce{Ba}\)
- \(\ce{Ba}\) \(<\) \(\ce{Sr}\) \(<\) \(\ce{Mg}\) \(<\) \(\ce{S}\) \(<\) \(\ce{Ar}\)
- \(\ce{Ba}\) \(<\) \(\ce{Mg}\) \(<\) \(\ce{S}\) \(<\) \(\ce{Sr}\) \(<\) \(\ce{Ar}\)
- \(\ce{Ar}\) \(<\) \(\ce{Sr}\) \(<\) \(\ce{Mg}\) \(<\) \(\ce{S}\) \(<\) \(\ce{Ba}\)
- \(\ce{Ba}\) \(<\) \(\ce{Mg}\) \(<\) \(\ce{Sr}\) \(<\) \(\ce{S}\) \(<\) \(\ce{Ar}\)
- Which of the following elements has the lowest first ionisation energy?
- \(\ce{Rb}\)
- \(\ce{Mo}\)
- \(\ce{Ag}\)
- \(\ce{Cs}\)
- Which of the following statements is true?
- First ionisation energy increases with increasing atomic radii
- First ionisation energy increases with decreasing atomic radii
- First ionisation energy of two \(d\) block elements located in the same period changes significantly
- First ionisation energy increases down a group
- Which of the following statements is incorrect regarding the electron affinity?
- Electron affinity is the energy change when an electron is added to a free atom in the gaseous phase
- Electron affinity becomes more negative when moving across a period
- Electron affinity increases down a group
- There is no clear trend in electron affinity as \(n\) increases
- Predict which of the following atoms has the most negative electron affinity.
- \(\ce{K}\)
- \(\ce{Ti}\)
- \(\ce{As}\)
- \(\ce{Br}\)
- Which of the following elements has the highest electronegativity?
- \(\ce{P}\)
- \(\ce{F}\)
- \(\ce{Cl}\)
- \(\ce{Br}\)
- As you move from top to bottom in a group of the periodic table
- Atomic radii decreases
- First ionisation energy increases
- Electronegativity decreases
- Electron affinity increases
- Arrange the following atoms in order of increasing electronegativity.
\(\ce{N}\), \(\ce{O}\), \(\ce{S}\), \(\ce{Se}\)
- \(\ce{Se}\) \(<\) \(\ce{S}\) \(<\) \(\ce{N}\) \(<\) \(\ce{O}\)
- \(\ce{Se}\) \(<\) \(\ce{N}\) \(<\) \(\ce{S}\) \(<\) \(\ce{O}\)
- \(\ce{Se}\) \(<\) \(\ce{S}\) \(<\) \(\ce{O}\) \(<\) \(\ce{N}\)
- \(\ce{O}\) \(<\) \(\ce{S}\) \(<\) \(\ce{N}\) \(<\) \(\ce{Se}\)
