States of matter
What is matter?
Matter is anything that has mass and occupies space. It includes things that can be seen (all living and non-living, natural and synthetic things) and cannot be seen to the naked eye (e.g. air and microorganisms).
Physical states of matter
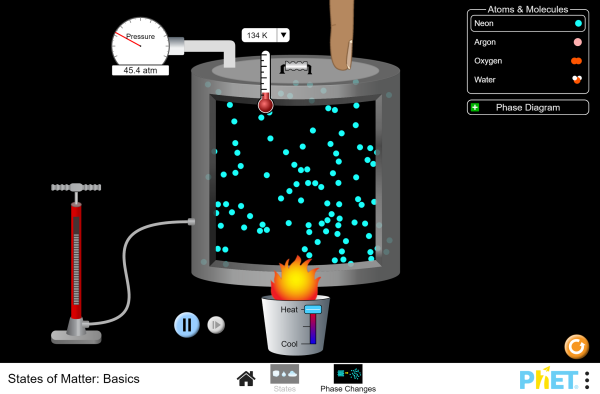
Matter exists in three physical states: solid, liquid and gas.
Solid
The particles in a solid are packed tightly with a regular pattern. Hence, they do not pass each other. Solids have a fixed shape and a fixed volume.
Liquid
The particles in a liquid are situated close together without a regular pattern. The distance between two particles is large enough for one particle to pass the other. Liquid takes the shape of the container - it has an indefinite shape. Liquid has a fixed volume.
Gas
The particles in a gas are separated without a regular pattern. Gas takes the shape of the container - it has an indefinite shape. Gas fills the space of the container - it has an indefinite volume.



Change of state
The conversion of a substance from one state to another is known as the change of state.
Examples:
- Melting of ice (solid to liquid).
- Boiling of water (liquid to gas).
- Freezing of water (liquid to solid).
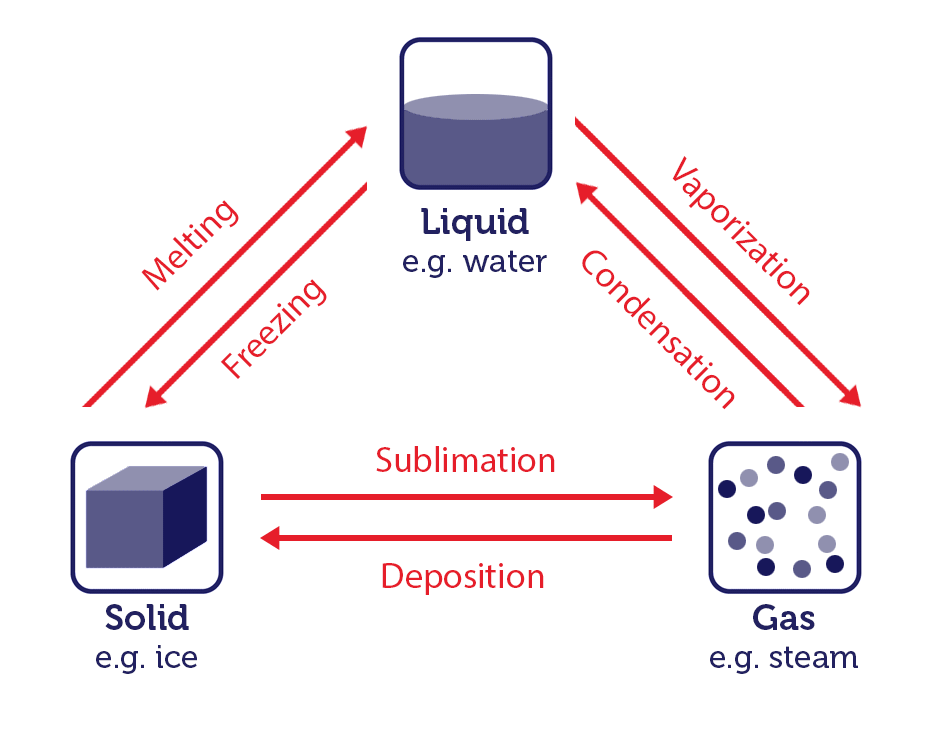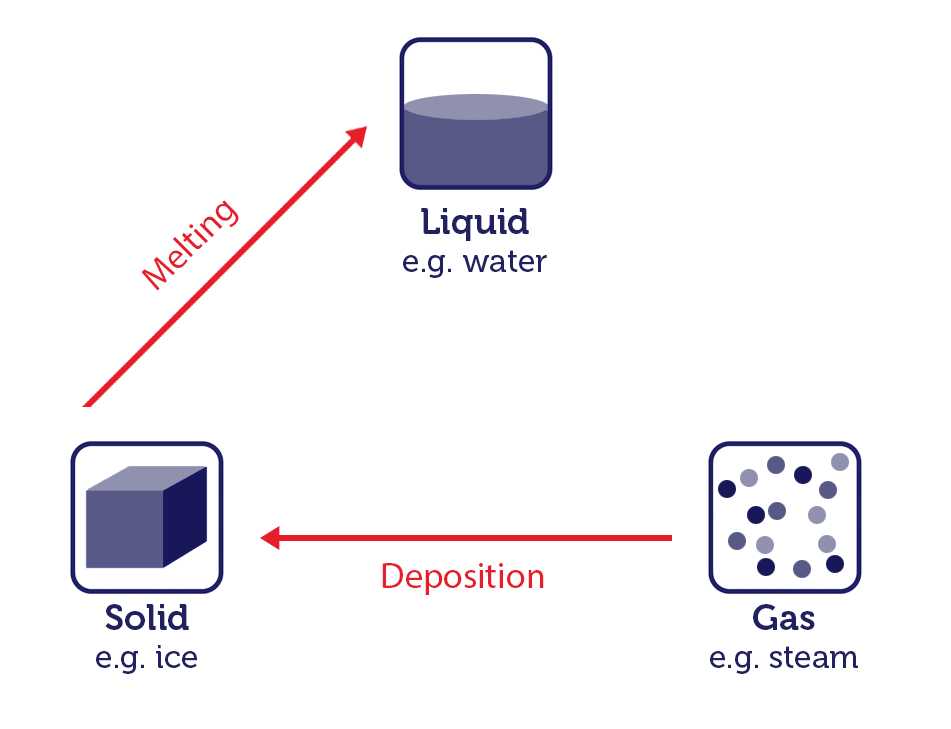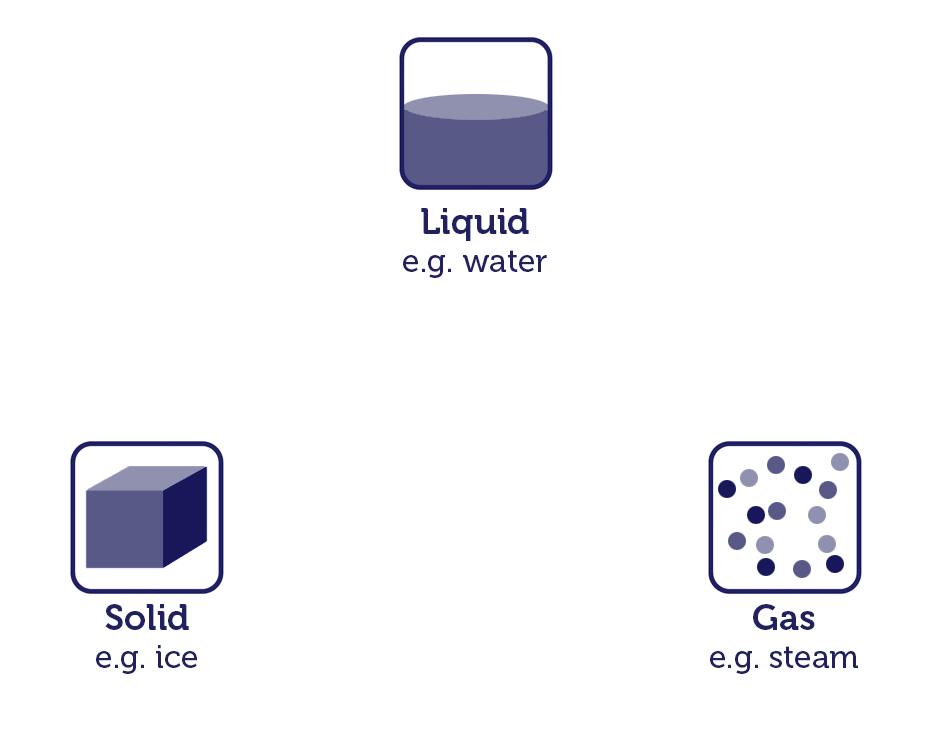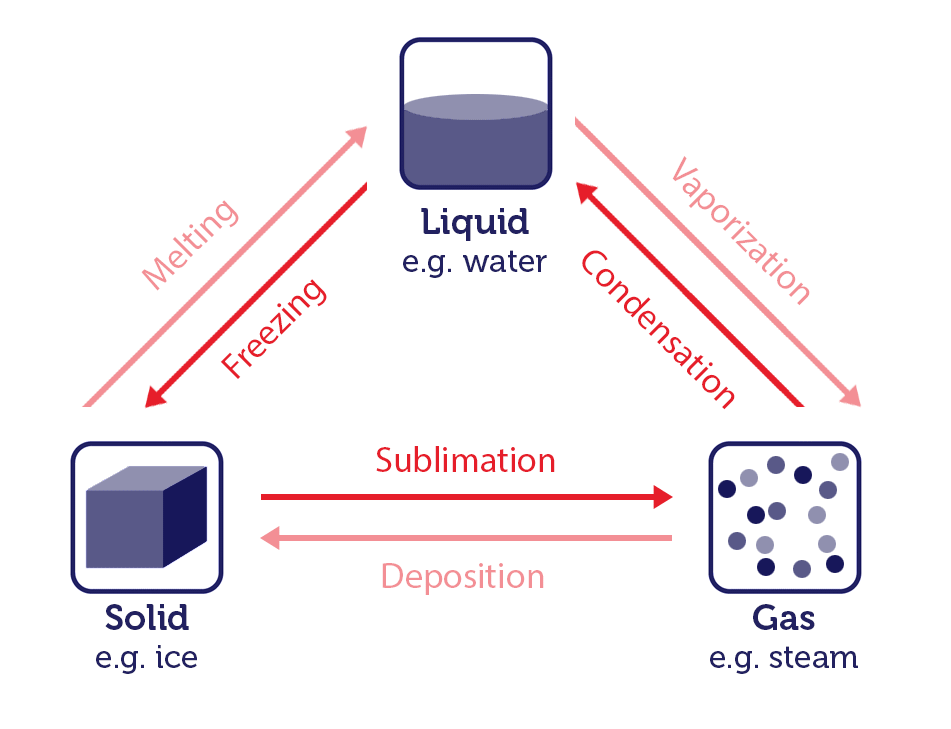
Water is one of the few substances which can exist in all three states: solid ice, liquid water, gaseous steam. State changes have different names depending on which states are involved:
- The change of solid to liquid is known as melting.
- The change of liquid to gas is known as vaporization.
- The change of gas to solid is known as deposition.
- The change of solid to gas is known as sublimation.
- The change of liquid to solid is known as freezing.
- The change of gas to liquid is known as condensation.
Properties of matter
Properties of matter can be classified into two types depending on whether the particular property is determined by changing the chemical identity or composition of the substance.
| Are the properties observed/measured without changing the chemical identity of the substance? | |
NO = chemical property |
YES = physical property |
| Flammability - when wood burns it changes to ashes | Shape |
|
Corrosion - rusting of iron objects after exposure to moist air for a long time |
Colour |
| Decomposition - hydrogen peroxide decomposes into water and oxygen in the presence of heat or light | Odour |
| Reactivity - potassium reacts with water | Hardness |
| Melting point | |
|
Boiling point |
|
| Physical state (liquid, solid, gas) | |
Changes in the matter
Changes in the matter are classified into two categories: physical change and chemical change.
Physical change
A physical change is when the substance changes its physical appearance or state without affecting the chemical identity. For example, the melting of ice to give water is a physical change because water only changes its physical state from solid to liquid without affecting its chemical identity.
Examples:
- the grating of a piece of cheese
- boiling of water
- sugar dissolving in water
Chemical change
A chemical change is a process in which the substance changes its chemical identity or composition. It involves the formation of new substances that have different properties compared to the original substance. For example, iron objects turn reddish-brown (known as rusting) when exposed to moist air for a long period is a chemical change because iron combines with oxygen and moisture from the air to form a new substance, rust.
Examples:
- sugar heated in a saucepan to make caramel
- burning of potassium in water
- milk turning sour
Classification of matter
The flowchart below shows how you can determine the classification of matter through a series of simple yes/no questions.
Classification of matter
Matter
Pure substance
e.g. Water, sugar, table salt
Chemical compound
Element
Mixture
e.g. Salt-sand mixture, sea water, concrete
Homogenous Mixture
Hetereogeneous mixture
Test your knowledge
Take the States of matter: Quiz
Use the States of Matter: Interactive